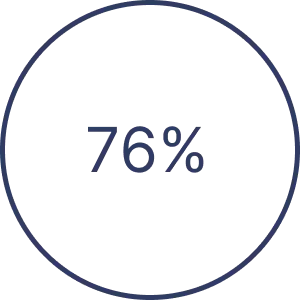
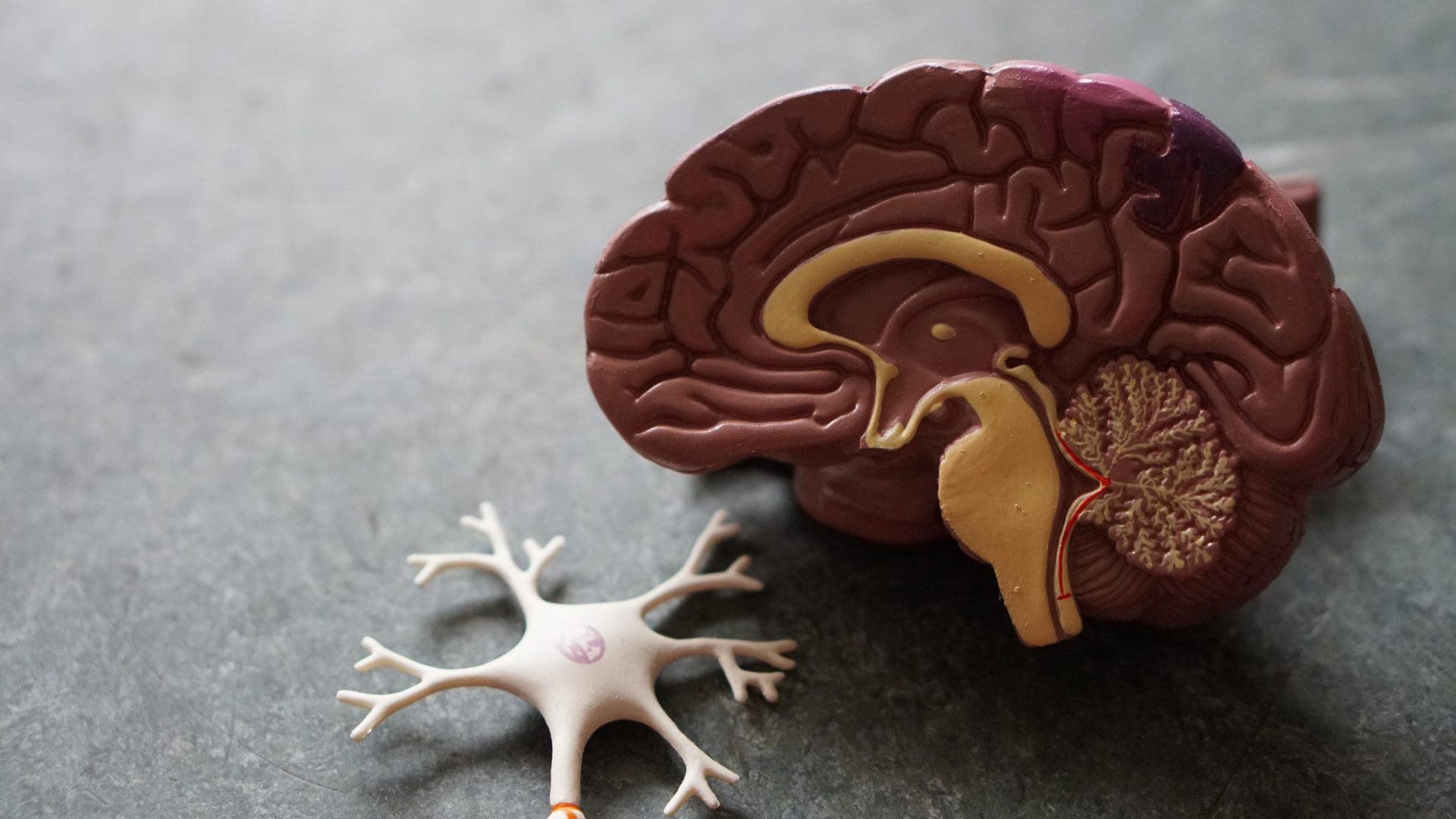
Discover mental exercises for mild cognitive impairment, including brain games, meditation, and physical activity, aimed…
read more
“I use OptoCeutics’ light everyday! Usually in the morning when I’m cooking or eating breakfast. It helps me have peace of mind as a preventative measure to an aging brain.”

“I feel a mood and energy boost after using my EVYLIGHT®.”

“Due to problems with my short-term memory in 2020, I acquired one of the first lamps that was produced. I used it for an hour a day, for approx. half a year. I can not say whether the use of the lamp is the reason for the small improvement in memory that I have experienced, but the use of the lamp is easy and comfortable – with a slightly meditative nature. The team at OptoCeutics is very enthusiastic and helpful.”

“Effective medical lamp which gives me more energy, better sleep and improves my mood. I can clearly feel the difference when I use it regularly!”

“It has been and is a very positive experience to use the lamp for 1 hour every day. I have gained more energy and joy in my daily life.”

“I always get my bank account number mixed up but lately I’ve been very surprised to remember it perfectly. It usually takes me at least three times to get it right, but now it’s BOOM, first try. I also tend to have a harder time with subtraction compared to addition, but my subtracting seems to be getting better. I really think it’s the machine that’s helping me. I use it every morning – meditation, reading, knitting, or watching TV.”